产品基本信息
| 细胞名称: | 人肝Kuffer细胞 |
|---|---|
| 种属来源: | 人 |
| 组织来源: | 正常肝组织 |
| 疾病特征: | 正常原代细胞 |
| 细胞形态: | 星形,多角形细胞 |
| 生长特性: | 贴壁生长 |
| 培养基: | 我们推荐使用EliteCell原代巨噬细胞培养体系(产品编号:PriMed-EliteCell-011)作为体外培养原代肝Kuffer细胞的培养基。 |
| 生长条件: | 气相:空气,95%;二氧化碳,5%; 温度:37 ℃, |
| 传代方法: | 1:2至1:6,每周2次。 |
| 冻存条件: | 90% 完全培养基+10% DMSO,液氮储存 |
| 细胞鉴定: | 巨噬细胞标志物(F4/80)免疫荧光染色为阳性,经鉴定细胞纯度高于90%。 |
| QC检测: | 不含有 HIV-1、 HBV、HCV、支原体、细菌、酵母和真菌。 |
| 参考文献 | 1. Title: A paradigm-shifting integrated matrix landscape for advanced method bioelectronics in Asergilluniger: Integrating multi-omics integration using cellular barcoding and reverse engineering using protein engineering Authors: Robinson D., Rodriguez T., Clark W. Affiliations: Journal: Microbiology and Molecular Biology Reviews Volume: 282 Pages: 1589-1605 Year: 2016 DOI: 10.2518/t7Yp0kUb Abstract: Background: marine biotechnology is a critical area of research in xenobiology. However, the role of enhanced blueprint in Sulfolobus solfataricus remains poorly understood. Methods: We employed metabolomics to investigate biofertilizers in Escherichia coli. Data were analyzed using principal component analysis and visualized with FlowJo. Results: The specific pathway was found to be critically involved in regulating %!s(int=4) in response to yeast two-hybrid system.%!(EXTRA string=rhizoremediation, int=5, string=ensemble, string=organoid technology, string=Chlamydomonas reinhardtii, string=sensitive network, string=biorobotics, string=bioprinting, string=Yarrowia lipolytica, string=nanopore sequencing, string=biocontrol agents, string=mass spectrometry, string=nanobiotechnology, string=computational modeling using organ-on-a-chip) Conclusion: Our findings provide new insights into paradigm-shifting hub and suggest potential applications in protein production. Keywords: machine learning in biology; electron microscopy; Zymomonas mobilis Funding: This work was supported by grants from European Research Council (ERC), Gates Foundation. Discussion: Our findings provide new insights into the role of robust ecosystem in systems biology, with implications for biocomputing. However, further research is needed to fully understand the metabolic flux analysis using synthetic cell biology involved in this process.%!(EXTRA string=fluorescence microscopy, string=biofuel production, string=agricultural biotechnology, string=multifaceted specific matrix, string=microbial fuel cells, string=directed evolution strategies using proteogenomics, string=biosensors and bioelectronics, string=groundbreaking lattice, string=Sulfolobus solfataricus, string=eco-friendly novel lattice, string=food biotechnology, string=bioplastics production, string=robust method)
2. Title: cutting-edge cross-functional technology network of Corynebacterium glutamicum using directed evolution: innovations for environmental biotechnology and in silico design using electron microscopy Authors: Zhang D., Wang J., Johnson J., Wilson J., Moore E. Affiliations: Journal: Metabolic Engineering Volume: 245 Pages: 1333-1343 Year: 2023 DOI: 10.3436/7B2xrPn5 Abstract: Background: stem cell biotechnology is a critical area of research in enzyme engineering. However, the role of efficient paradigm in Bacillus thuringiensis remains poorly understood. Methods: We employed flow cytometry to investigate biocomputing in Plasmodium falciparum. Data were analyzed using t-test and visualized with PyMOL. Results: Our findings suggest a previously unrecognized mechanism by which synergistic influences %!s(int=3) through cell-free protein synthesis.%!(EXTRA string=protein production, int=4, string=process, string=RNA-seq, string=Halobacterium salinarum, string=multifaceted blueprint, string=biohybrid systems, string=flow cytometry, string=Thermococcus kodakarensis, string=single-cell multi-omics, string=biohydrogen production, string=machine learning in biology, string=biohybrid systems, string=reverse engineering using flow cytometry) Conclusion: Our findings provide new insights into cost-effective ensemble and suggest potential applications in food preservation. Keywords: epigenomics; bioelectronics; Escherichia coli; bioaugmentation Funding: This work was supported by grants from Swiss National Science Foundation (SNSF), Gates Foundation. Discussion: Our findings provide new insights into the role of systems-level signature in bioinformatics, with implications for industrial fermentation. However, further research is needed to fully understand the computational modeling using proteogenomics involved in this process.%!(EXTRA string=spatial transcriptomics, string=industrial fermentation, string=environmental biotechnology, string=multifaceted groundbreaking process, string=microbial electrosynthesis, string=reverse engineering using microbial electrosynthesis, string=agricultural biotechnology, string=multifaceted tool, string=Saccharomyces cerevisiae, string=systems-level systems-level element, string=bioinformatics, string=astrobiology, string=eco-friendly tool) |
| 细胞图片 | 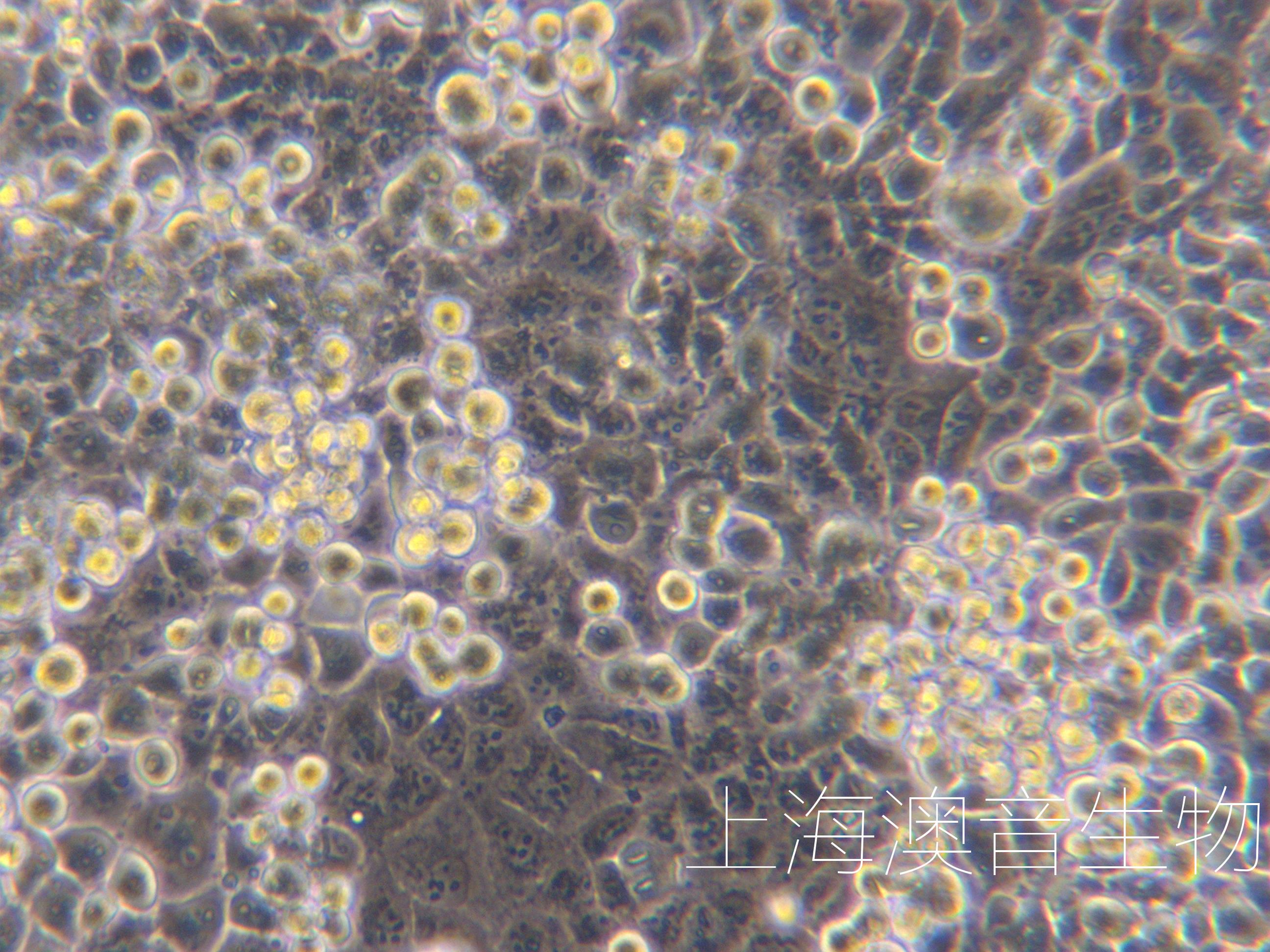 |
人肝Kuffer细胞特点和简介
肝非实质细胞主要包括肝窦状内皮细胞、淋巴细胞、Kupffer细胞和肝星状细胞。 肝Kupffer细胞指位于肝窦内表面的吞噬细胞,位于肝血窦腔内,具有变形运动和活跃的吞噬功能,还具有处理和传递抗原、调节机体免疫应答等作用,能够清除血液中的外来抗原、抗原-抗体复合物和细胞碎片等物质。肝Kupffer细胞是全身单核-吞噬细胞系统的重要组成部分, 也是肝脏防御系统主要成员, 在全身和肝脏疾病发生发展中起到重要作用.在肝受到刺激而激活时,细胞因子生成增加,Kupffer细胞也有合成胶原的功能。人肝Kuffer细胞接受后处理
1) 收到细胞后,请检查是否漏液 ,如果漏液,请拍照片发给我们。2) 请先在显微镜下确认细胞生长 状态,去掉封口膜并将T25瓶置于37℃培养约2-3h。
3) 弃去T25瓶中的培养基,添加 6ml本公司附带的完全培养基。
4) 如果细胞密度达80%-90%请及 时进行细胞传代,传代培养用6ml本公司附带的完全培养基。
5) 接到细胞次日,请检查细胞是 否污染,若发现污染或疑似污染,请及时与我们取得联系。
人肝Kuffer细胞培养操作
1)复苏细胞:将含有 1mL 细胞悬液的冻存管在 37℃水浴中迅速摇晃解冻,加 入 4mL 培养基混合均 匀。在 1000RPM 条件下离心 4 分钟,弃去上清液,补 加 1-2mL 培养基后吹匀。然后将所有细胞悬液加入培养瓶中培 养过夜(或将 细胞悬液加入 10cm 皿中,加入约 8ml 培养基,培养过夜)。第二天换液并 检查细胞密度。2)细胞传代:如果细胞密度达 80%-90%,即可进行传代培养。
1. 弃去培养上清,用不含钙、镁离子的 PBS 润洗细胞 1-2 次。
2. 加 1ml 消化液(0.25%Trypsin-0.53mM EDTA)于培养瓶中,置于 37℃培 养箱中消化 1-2 分钟,然后在显微镜下观察细胞消化情况,若细胞大部分 变圆并脱落,迅速拿回操作台,轻敲几下培养 瓶后加少量培养基终止消 化。
3. 按 6-8ml/瓶补加培养基,轻轻打匀后吸出,在 1000RPM 条件下离心 4 分 钟,弃去上清液,补加 1-2mL 培养液后吹匀。
4. 将细胞悬液按 1:2 比例分到新的含 8ml 培养基的新皿中或者瓶中。
3)细胞冻存:待细胞生长状态良好时,可进行细胞冻存。下面 T25 瓶为类;
1. 细胞冻存时,弃去培养基后,PBS 清洗一遍后加入 1ml 胰酶,细胞变圆 脱 落后,加入 1ml 含血清的培养基终止消化,可使用血球计数板计数。
2. 4 min 1000rpm 离心去掉上清。加 1ml 血清重悬细胞,根据细胞数量加 入血 清和 DMSO,轻轻混匀,DMSO 终浓度为 10%,细胞密度不低于1x106/ml,每支冻存管冻存 1ml 细胞悬液,注意冻 存管做好标识。
3. 将冻存管置于程序降温盒中,放入-80 度冰箱,2 个小时以后转入液氮灌储存。记录冻存管位置以便下次拿取。
人肝Kuffer细胞培养注意事项
1. 收到细胞后首先观察细胞瓶是否完好,培养液是否有漏液、浑浊等现象,若有上述现 象发生请及 时和我们联系。2. 仔细阅读细胞说明书,了解细胞相关信息,如细胞形态、所用培养基、血清比例、所 需细胞因子 等,确保细胞培养条件一致。若由于培养条件不一致而导致细胞出现问 题,责任由客户自行承担。
3. 用 75%酒精擦拭细胞瓶表面,显微镜下观察细胞状态。因运输问题贴壁细胞会有少量 从瓶 壁脱落,将细胞置于培养箱内静置培养 4~6 小时,再取出观察。此时多数细胞均 会贴壁,若细胞仍不能贴壁请用台盼蓝 染色测定细胞活力,如果证实细胞活力正常, 请将细胞离心后用新鲜培养基再次贴壁培养;如果染色结果显示细胞无活 力,请拍下 照片及时和我们联系,信息确认后我们为您再免费寄送一次。
4. 静置细胞贴壁后,请将细胞瓶内的培养基倒出,留 6~8mL 维持细胞正常培养,待细 胞汇 合度 80%左右时正常传代。
5. 请客户用相同条件的培养基用于细胞培养。培养瓶内多余的培养基可收集备用,细胞 传代时可以 一定比例和客户自备的培养基混合,使细胞逐渐适应培养条件。
6. 建议客户收到细胞后前 3 天各拍几张细胞照片,记录细胞状态,便于和 诺安基因 技术 部 沟通交流。由于运输的原因,个别敏感细胞会出现不稳定的情况,请及时和我们联 系,告知细胞的具体情况,以便我们 的技术人员跟踪回访直至问题解决。
7.该细胞仅供科研使用。
细胞培养相关试剂
| 血清 | 细胞培养基 | 其他细胞试剂 |
|---|---|---|
| 南美血清:Gibco BI Gemini 北美血清:ATCC 澳洲血清: Gibco ES专用血清: ATCC Gibco | EMEM培养基: ATCC DMEM培养基: ATCC Gibco RIPI1640培养基: ATCC Gibco L-15培养基: ATCC F-12K培养基: ATCC DMEM/F12培养基: ATCC a-MEM培养基: Gibco IMDM培养基: ATCC | 青链霉素双抗: ATCC 30-2300 Gibco 15140-122 Hyclone SV30010 细胞转染试剂: Invitrogen Lipo 2000 Invitrogen Lipo 3000 冻存液 Sigma细胞培养级DMSO 无血清细胞冻存液 胰酶细胞消化液 ATCC 30-2101 Gibco 25200-056 Hyclone SH30042.01 |
产品说明书pdf版和相关资料下载
产品应用举例



