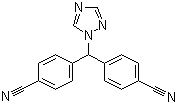
Grade: Pharmaceutical Grade
Factory Location: Lianyungang,Jiang su,China
Main Sales Markets: North America,Central/South America,Western Europe,Eastern Europe,Australasia,Asia,Middle East,Africa
Sample Provided: no
Letrozole is an oral non-steroidal aromatase inhibitor for the treatment of hormonally-responsive breast cancer after surgery.
Letrozole API has Chinese local GMP certificate and CEP certificate. It also has been approved by US-FDA and Japanese PMDA.
Letrozole Tablets have got Chinese local GMP certificate and EU GMP, and have MA issued from US-FDA, Japanese PMDA. It was approved by EU and launched in European market in 2012, approved by US FDA and Launched in American market in May, 2013 and approved to get launched in the Japanese market in 2015.
Currently, the sales of Letrozole FDF and API have covered over 10 countries in Europe, America and Asia.