Grade: Pharmaceutical Grade
Factory Location: zhejiang
Main Sales Markets: Asia,Africa
Monthly Production Capacity: 1t
Contract Manufacturing: CRO,CMO
Packaging Information: 1kg/Aluminum drum
Delivery Lead Time: 2-3Week
Sample Provided: no
Payment Terms: DP/DA/TT/LC
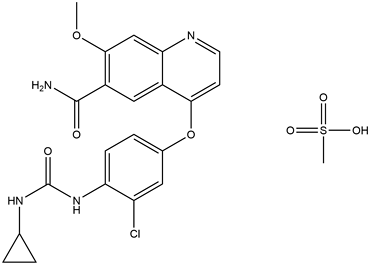
Lovatinib is a multi-target tyrosine kinase inhibitor, which may act on a variety of cancers, so Eisai is still studying Lovatinib for other cancers, such as hepatocellular carcinoma, non-small cell lung cancer, melanoma, breast cancer, lymphoma, ovarian cancer, etc. There were 62 clinical studies related to lovatib chemicalbookil. In July 2017, Eisai submitted a marketing application for levatinib as a first-line treatment for advanced hepatocellular carcinoma to the US FDA and the European Medicines Agency. In clinical trials, levatinib was superior to sorafenib, the current standard treatment for advanced hepatocellular carcinoma.