Grade: Pharmaceutical Grade
Factory Location: Shandong
Main Sales Markets: North America,Central/South America,Western Europe,Eastern Europe,Australasia,Asia,Middle East,Africa
Monthly Production Capacity: 100kg
Contract Manufacturing: CRO,CMO
Packaging Information: 5kg/drum
Delivery Lead Time: 15days
Sample Provided: yes
Payment Terms: L/C
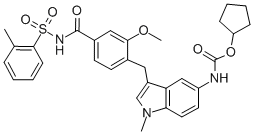
Revefenacin is a novel biphenyl carbamate tertiary amine agent that belongs to the family of the long-acting muscarinic antagonists (LAMA). The labile primary amide in the structure produces a "soft-drug" site that allows rapid systemic clearance and minimizing of the systemically mediated adverse reactions. The LAMA group falls into a parent category known as long-acting inhaled bronchodilators and this type of agents are recommended as a maintenance therapy for chronic obstructive pulmonary disease (COPD). From the LAMA group, revefenacin is the first once-daily nebulized LAMA treatment. It was developed by Theravance Biopharma and FDA approved on November 9, 2018.