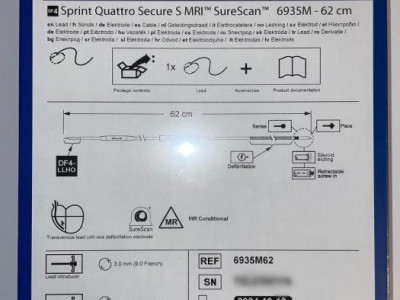
Sprint Quattro Family of Leads Indications Medtronic Sprint Quattro™ leads are intended for pacing and sensing and/or defibrillation. Defibrillation leads have application for patients for whom implantable cardioverter defibrillation is indicated. The Sprint Quattro™ MRI SureScan™ leads (which include specified lengths of Models 6935, 6935M, 6947, and 6947M) are part of a Medtronic SureScan™ ICD or CRT-D system. Consult individual lead model technical manuals for more detail. A complete SureScan™ defibrillation or CRT-D system is required for use in the MR environment and includes a Medtronic SureScan™ device connected to Medtronic SureScan™ leads. Contraindications The Sprint Quattro™ leads are contraindicated: § For the sole use of detection and treatment of atrial arrhythmias § In patients with tricuspid valvular disease and/or patients with mechanical tricuspid heart valves § For patients with transient ventricular tachyarrythmias due to reversible causes (drug intoxication, electrolyte imbalance, sepsis, hypoxia) or other factors (myocardial infarction, electric shock) § In patients for whom a single dose of 1.0 mg of dexamethasone acetate and dexamethasone sodium phosphate may be contraindicated